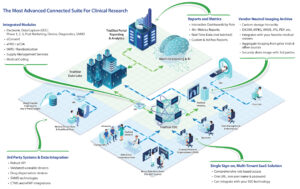
eClinical Technology
Selecting the Right EDC: A Critical Decision for Clinical Trial Success In the ever-evolving landscape of clinical trials, the choice of an Electronic Data Capture (EDC) technology vendor is more than a mere procedural step; it’s a pivotal decision that can significantly impact the success of your clinical research. Whether you’re embarking on your first…
Read MoreYou should not have to compromise your study design simply because your eClinical Technology does not support your unique study requirements. At TrialStat we do things differently. We believe that you should not have to compromise and to make that a reality, we specialize in Custom Validated Development. Whether it’s a small change in workflow,…
Read MoreTrialStat’s eConsent platform was designed and developed to solve the most pressing trial enrolment and protocol compliance problems. With a completely customizable workflow, content templates, multimedia capabilities, and multilingual options, Sponsors and CROs have the tools they need to create an immersive eConsent process for trial participants. Our eConsent module is fully integrated with our…
Read MoreThe landscape of clinical research is constantly evolving, and businesses need to keep up with the latest technology to remain competitive. However, investing in eClinical technology is not just about finding a solution for current needs, it’s equally important to think ahead and ensure that your technology investment can adapt and scale to meet future…
Read MoreFor 2023, we’re taking our blog in a new direction with more content directly from our team, more detailed information for our readers answering common questions we get, and helpful expert information and ideas you can use. We hope you enjoy this new approach. For this week, we’re covering an introduction (or refresher) to TrialStat…
Read More